Plant DNA extraction is a crucial technique in molecular biology. It allows scientists to study plant genetics, evolution, and breeding. Understanding this process can help researchers in conservation and agriculture.
Various methods exist for effective Plant DNA Extraction, each with its own pros and cons. For example, some methods may yield high-quality DNA but are time-consuming. Others are quick but may compromise DNA integrity. These trade-offs often need careful evaluation.
To help navigate this landscape, we’ve compiled a list of the top 10 methods. Each method has unique characteristics that can influence your study's outcome. Choosing the right one is essential, yet it can be challenging. This guide serves as a resource to enhance your understanding and implementation of plant DNA extraction techniques.

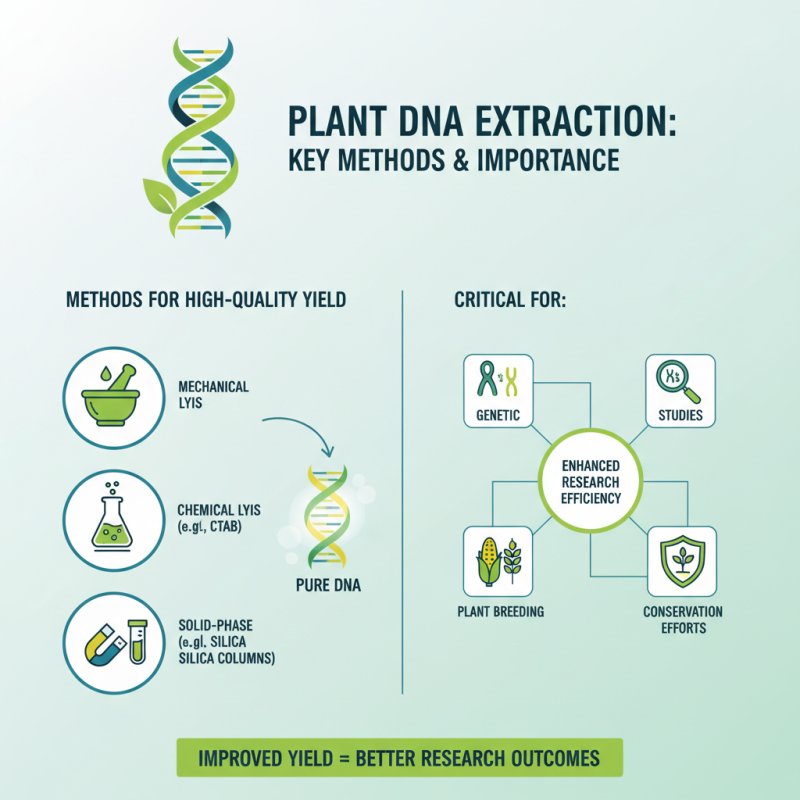
Plant DNA extraction is a critical technique in molecular biology. Understanding various methods can significantly enhance the efficiency of research. Recent studies show that proper extraction methods improve the yield of high-quality DNA. This has become increasingly vital for genetic studies, plant breeding, and conservation efforts.
Modern methods such as CTAB, silica gel, and magnetic bead-based techniques are widely used. Each of these techniques has distinct advantages and challenges. A report from the International Society for Molecular Plant-Microbe Interactions indicates that CTAB extraction often results in more intact samples. However, it can be time-consuming. Meanwhile, magnetic bead methods provide cleaner DNA in less time but may require specialized equipment.
The choice of method depends on various factors, such as the type of plant tissue and the intended application. Researchers should carefully evaluate their needs. In some cases, a less effective method may hinder progress. The ongoing challenge lies in balancing efficiency, cost, and quality. As the field evolves, refining these methods will be crucial for future advancements in plant genomics.
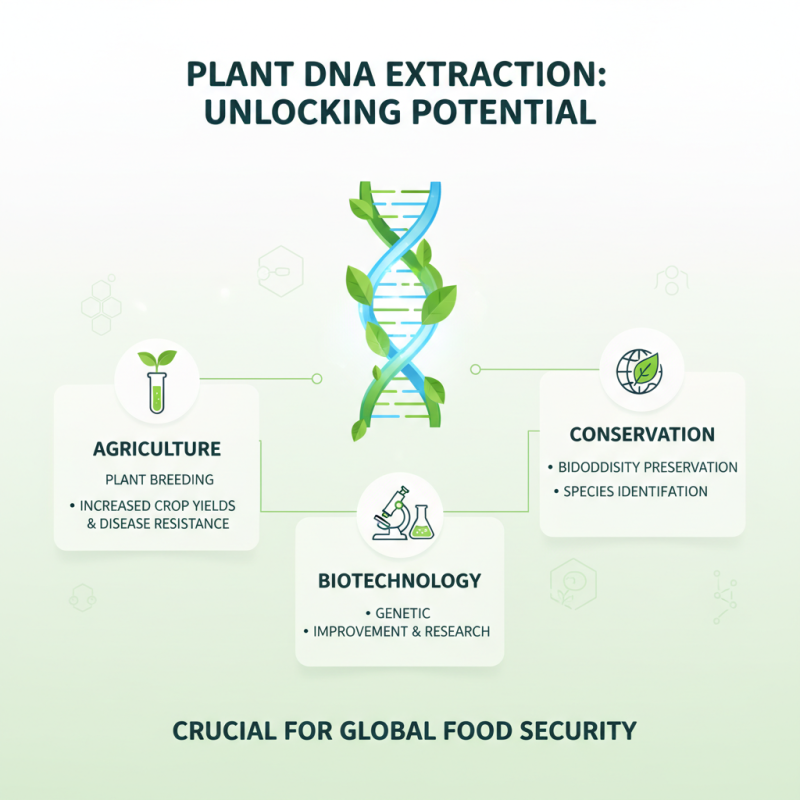
Understanding the importance of plant DNA extraction lies in its far-reaching applications in agriculture, biotechnology, and conservation. According to recent studies, effective DNA extraction can enhance plant breeding programs, leading to increased crop yields and disease resistance. As global food demand rises, optimizing these techniques becomes crucial.
Plant DNA extraction methods are vital for research in genetics and genomics. They help identify traits and support the development of genetically modified organisms (GMOs). The accuracy of these techniques impacts research outcomes. For instance, inefficient extraction can lead to contamination, yielding unreliable results. This aspect highlights the need to refine methodologies continually.
Tips: Focus on utilizing specific extraction protocols tailored for your plant species. This practice may improve yield and purity of DNA. It's also essential to be aware of the limitations of each method. Many researchers underestimate factors like sample quality and environmental conditions, which can significantly affect final results. Engaging in thorough troubleshooting is key.
When isolating plant DNA, several techniques stand out. One common method is the CTAB protocol. This technique uses a detergent to break down cell membranes. It effectively separates the DNA from other cellular components. Another popular approach is the use of silica columns. These columns bind DNA and allow for easy purification. The process is straightforward and often used in labs.
Tips: Always use fresh plant samples for the best results. Older samples may degrade DNA quality. Carefully optimize the pH levels in your extraction buffer. Too high or low a pH can affect yield.
Another technique worth mentioning is the NaOH method. It involves alkaline lysis for cell disruption. This method is quick but can lead to contamination if not executed properly. Adjusting the concentration of NaOH is crucial here.
Tips: Ensure all your reagents are pure and free from contaminants. Small impurities can greatly alter your results. Keep your workspace clean and organized to avoid mix-ups.
The CTAB method is a widely used technique for DNA extraction from plant tissues. It offers a reliable way to isolate high-quality DNA. This method uses cetyltrimethylammonium bromide (CTAB) to precipitate polysaccharides and proteins, which can interfere with DNA extraction. In a comparative study, it was found that the CTAB method yields more DNA compared to other methods, making it a preferred choice for researchers.
To perform this method, start by grinding fresh plant tissue in liquid nitrogen. Then, add the CTAB extraction buffer, which often contains salt and buffer agents. Incubate the mixture at 65°C for 30 minutes. This step helps to break down cell walls and membranes. After cooling, chloroform and isoamyl alcohol are added for phase separation. This is often the trickiest part, as not using the correct proportions can lead to poor separation.
Finally, precipitate the DNA using isopropanol. It’s crucial to wash the DNA with ethanol to remove any residual contaminants. Results may vary based on plant species and tissue type. Some may struggle to extract quality DNA, reflecting the importance of optimizing each step in this procedure. Reports indicate that approximately 20% of samples may not yield the desired results. This indicates the necessity for careful execution and sometimes repeated attempts.
When selecting a method for plant DNA extraction, efficiency and yield are crucial. Each technique has its pros and cons. Some methods are faster but may yield less DNA. Others are more reliable but time-consuming. It's essential to evaluate these factors based on your specific plant samples.
One common method is the CTAB extraction. It is known for producing high-quality DNA. However, it can be labor-intensive and requires careful handling of reagents. Results may vary depending on the type of plant. Always pay attention to details. For example, younger leaves often provide better yields.
Tips: Optimize your extraction protocol for different plant types. Sometimes, a simpler method may suffice for certain samples. Don't hesitate to adjust the extraction time or the amount of starting material. Testing multiple methods can reveal the most effective approach for your needs. This trial-and-error process can be both enlightening and frustrating. It's part of the journey in discovering what works best for your research.
| Method | Efficiency (%) | Yield (µg/g) | Time (minutes) | Complexity Level |
|---|---|---|---|---|
| CTAB Method | 85 | 200 | 60 | Moderate |
| SDS Method | 80 | 180 | 50 | Easy |
| CTAB-PEG Method | 90 | 250 | 75 | High |
| Alkaline Lysis | 75 | 150 | 45 | Moderate |
| DNeasy Plant Mini Kit | 95 | 300 | 30 | Easy |
| Magnetic Bead Method | 88 | 220 | 40 | Moderate |
| Phenol-Chloroform Extraction | 82 | 200 | 90 | High |
| Salting-Out Method | 78 | 160 | 55 | Moderate |
| GeneJET Plant Genomic DNA Purification Kit | 94 | 290 | 35 | Easy |
| NucleoSpin Plant II Kit | 91 | 280 | 30 | Easy |